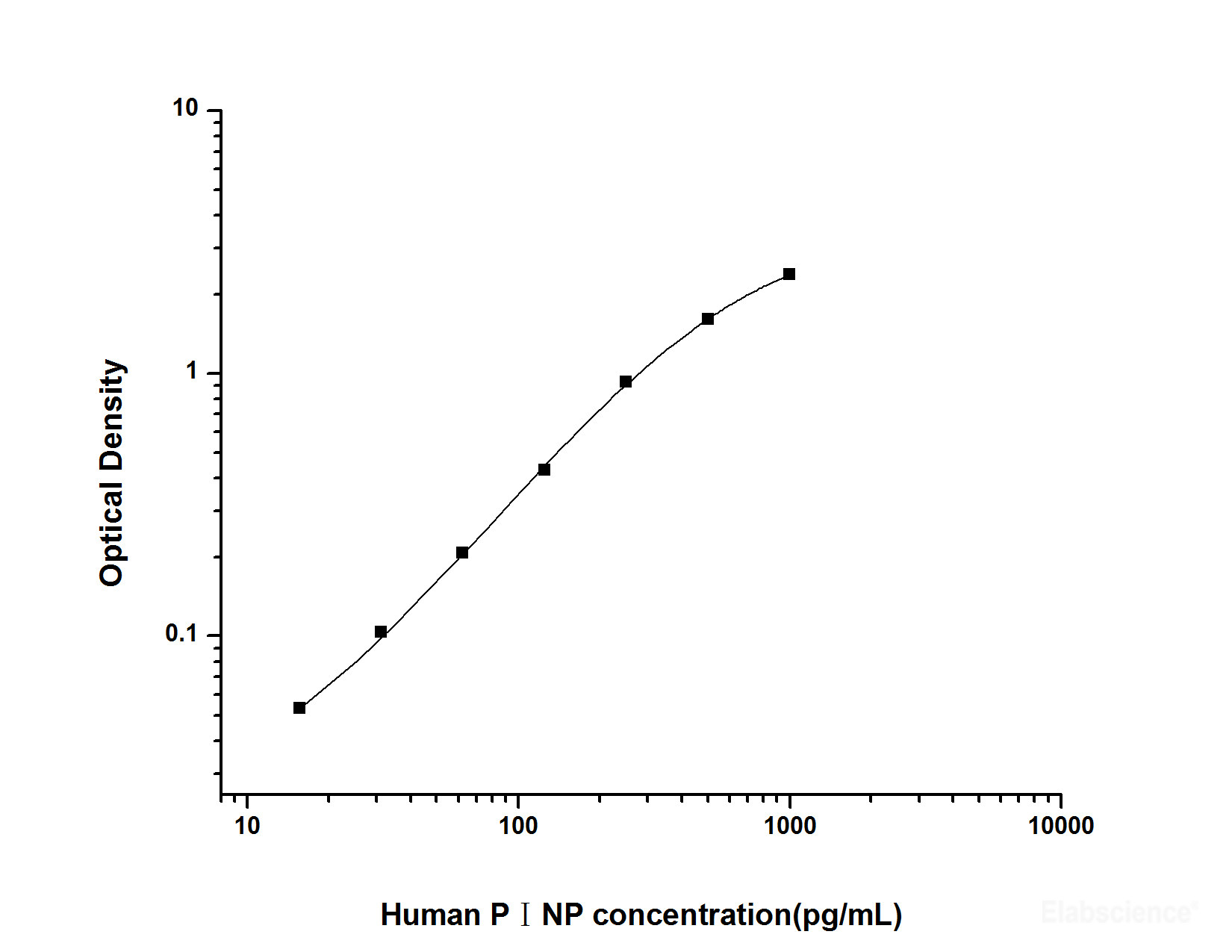
Human PⅠNP(Procollagen Ⅰ N-Terminal Propeptide) ELISA Kit
E-EL-H0185
| Size: | Price: | $609 | |
| Qty: |
-
+
|
Lead Time: Order now, ship in 3 days

Product Details
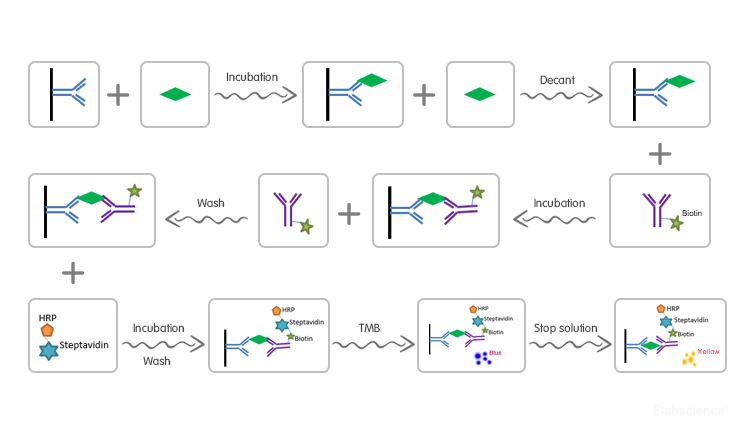
| Assay type | Sandwich-ELISA |
| Assay time | 3.5h |
| Detection range | 15.63-1000 pg/mL |
| Sensitivity | 9.38 pg/mL |
| Sample type &Sample volume | serum, plasma and other biological fluids; 100μL |
| Specificity | This kit recognizes Human PⅠNP in samples. No significant cross-reactivity or interference between Human PⅠNP and analogues was observed. |
| Reproducibility | Coefficient of variation is < 10% |
| Application | This ELISA kit applies to the in vitro quantitative determination of Human PⅠNP concentrations in serum, plasma and other biological fluids. |
Test Principle